What We Do
What is the GMDN?
The Global Medical Device Nomenclature (GMDN) is the world’s foremost standard for naming, defining, and categorising medical devices.
Anyone can register for free as a member on the GMDN website to access and use any GMDN Term.
Established in 1991, the first international workshop on medical device nomenclatures was held to create the GMDN. Initially started by the European Standards Organisations (CEN) and later supported by the Global Harmonisation Task Force (now the International Medical Device Regulators Forum – IMDRF) to help accelerate the harmonisation of medical device regulation globally. The GMDN is recommended by the IMDRF and is now used by circa 70 national medical device regulators across the globe.
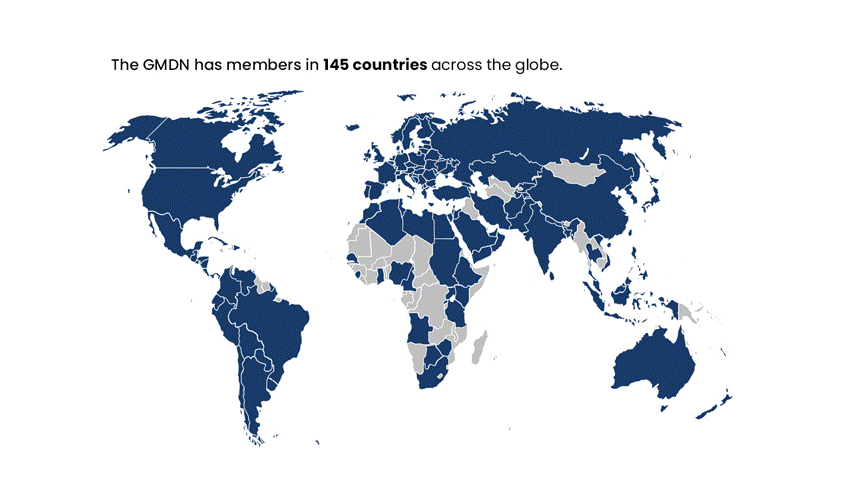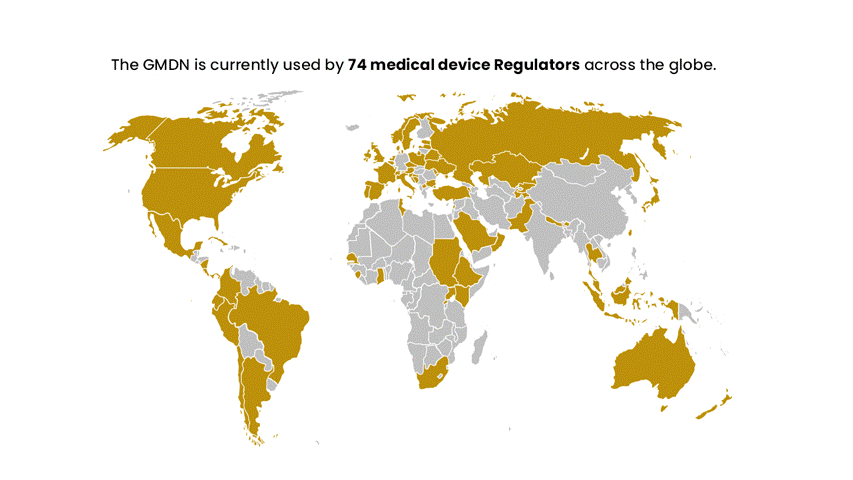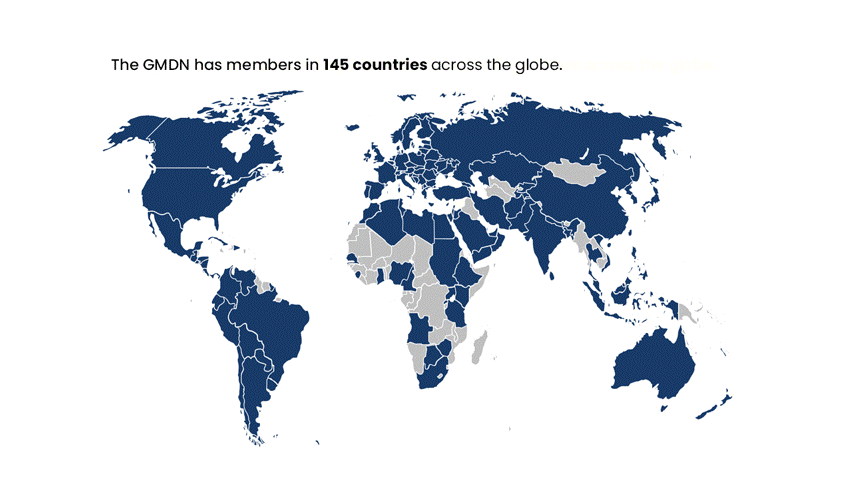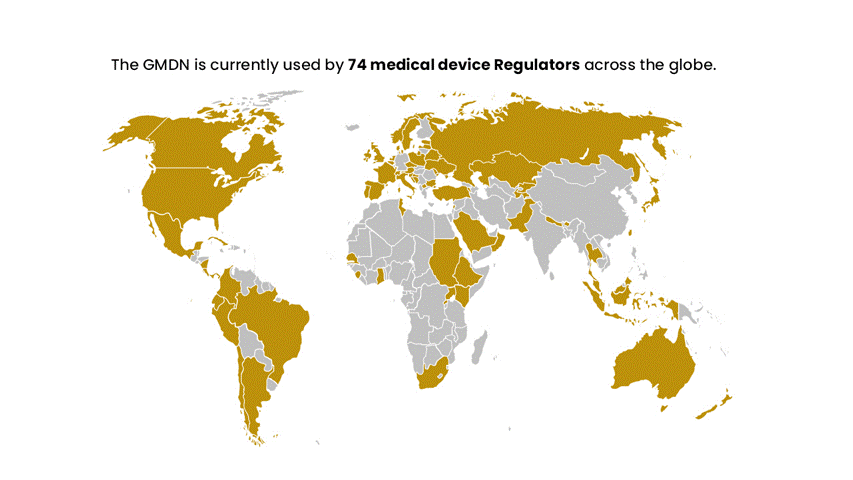
Global Reach and Inclusivity: GMDN is trusted in over 140 countries and is designed to serve regulators, manufacturers, and healthcare providers worldwide, not limited to the priorities of any single region.
Multi-Hierarchical Classification: Unlike single-hierarchical systems (such as EMDN or UNSPSC), GMDN’s multi-hierarchical approach allows for mutually-exclusive Terms and clinically relevant definitions, supporting more precise device identification and categorisation.
Real-Time Updates: GMDN is updated continuously, ensuring that new technologies and regulatory requirements are reflected promptly. Other systems may only update annually or bi-weekly, which can delay the adoption of innovations.
Clinically Relevant Definitions: GMDN Terms include definitions with intended use and important clinical attributes, supporting regulatory submissions, surveillance, and safety signal detection. Some other nomenclatures lack clinically relevant definitions or are less granular.
Flexible Access Models: GMDN offers both free of charge and paid options, funded by registered members, making it accessible to a wide range of stakeholders.
The full GMDN is available for free to medical device regulators and healthcare providers.
What is the GMDN Agency?
The GMDN Agency is structured as a non-profit, independent charity, responsible for the ongoing maintenance of the GMDN Database. The GMDN Agency is a wholly UK based organisation with no subsidiaries, franchises or licenced entities outside of the UK. Governed by a Board of Trustees representing the medical device sector from around the world. This ensures that the GMDN Agency does not prioritise any single jurisdiction or region but instead provides equal access and service to all stakeholders globally.
Independence is maintained through regular scrutiny by advisory groups such as the Technical Advisory Group (TAG) and the Authorities Strategic Advisory Group (ASAG), which include representatives from multiple countries and regulatory bodies. This governance model prevents regional bias and supports the needs of a diverse, International user base.
The GMDN Agency offers timely and responsive membership services, GMDN Term support, bespoke training sessions, and more, which are unique to GMDN compared to other nomenclatures.
Read more about our vision and objectives in our 2026-2030 – 5 year strategy.
You can download our latest annual report from the Charities Commission website.
-
Our Vision
To unify the global medical device community through a common nomenclature, underpinned by robust terminology, taxonomy and supporting data infrastructure and services – driving harmonisation, reducing regulatory burden, and enabling equitable access to safe and effective technologies.
-
Our Mission
We deliver a globally harmonised medical device nomenclature that safeguards public health by enabling seamless data interoperability, traceability, and informed decision-making across regulatory and healthcare systems.
How do we update the GMDN?
The GMDN Agency employs a team of expert Term Developers who analyse new products and technologies in the MedTech industry to create concise and accurate descriptions. If a product is unique, a new GMDN Term is created, but if it’s a variant of an existing product, an amendment to the Term Name or Definition may be made. Occasionally, a review of GMDN Terms may be needed to determine if they reflect the current devices on the market.
The decision to amend or create a new Term is made in consultation with manufacturers, sometimes with regulators’ input, and is dependent on the complexity of the product and the impact of any changes on the GMDN Database.
The GMDN Agency aims to complete the processes as soon as possible to make the updated information available to manufacturers and regulators.
GMDN global use
In 1991 the first international workshop on medical device nomenclatures was held to create the GMDN. Initially started by the European Standards Organisations (CEN) and later supported by the Global Harmonisation Task Force (now the International Medical Device Regulators Forum – IMDRF) to help accelerate the harmonisation of medical device regulation globally.
The GMDN is recommended by the IMDRF and is now used by circa 70 national medical device regulators across the globe. The GMDN is managed by the GMDN Agency, a registered charity, which has a Board of Trustees representing regulators and industry.
The GMDN is currently used by around 70 medical device Regulators across the globe and has members in around 140 countries worldwide.